Covid-19 Test
Due to the highly contagious nature and global health crisis, SARS-CoV-2 has been designated as a pandemic by the World Health Organization (WHO) and continues to have devastating impacts on healthcare systems and the world economy including the U.S. To effectively end the SARS-CoV-2 pandemic, systematic screening and detection of both clinical and asymptomatic COVID-19 cases is critical.
Ubase Life & BioHealth Inc. acts as an Authorized distributor for Celltrion DiaTrust COVID-19 Ag Home Test Kits
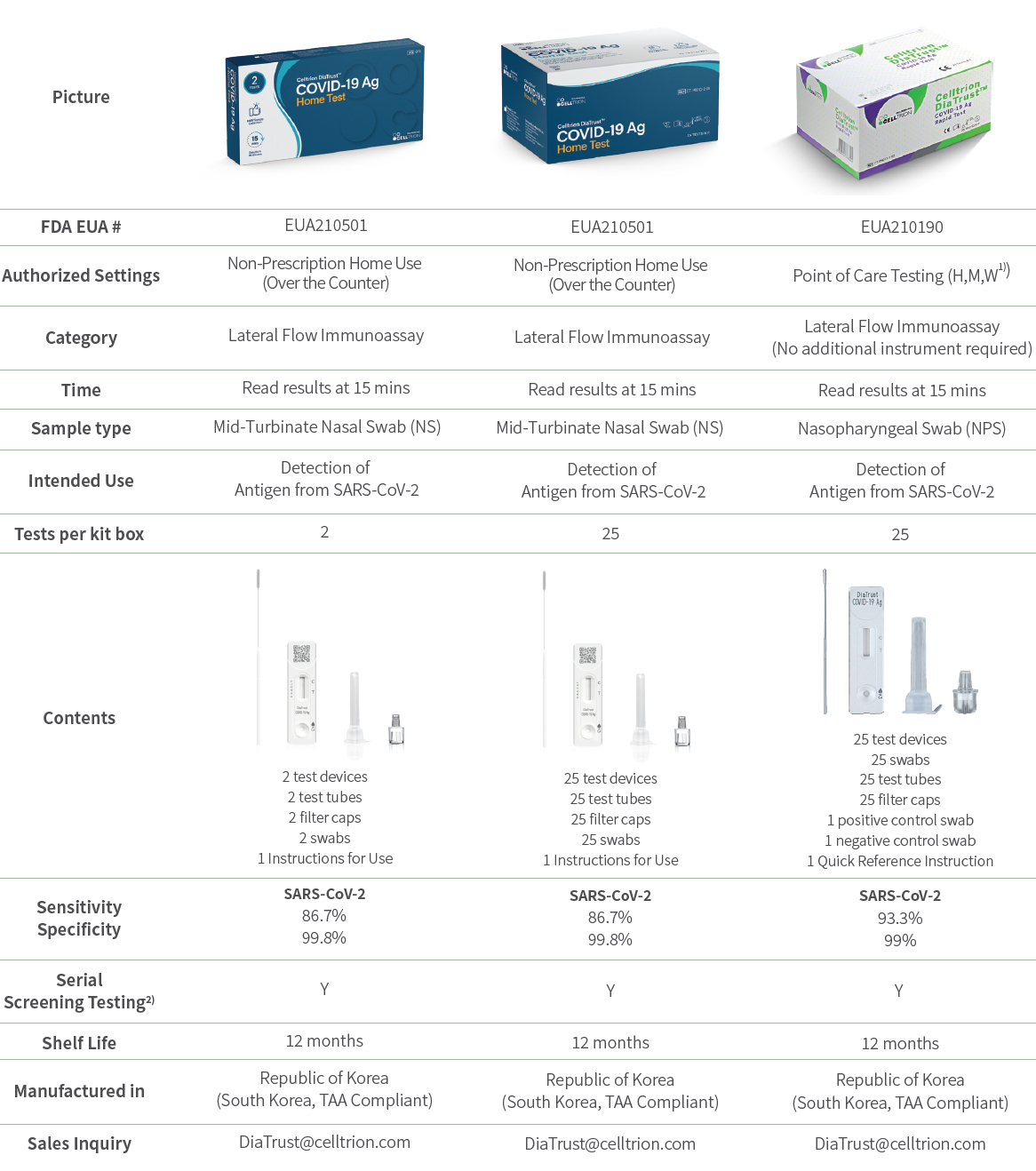
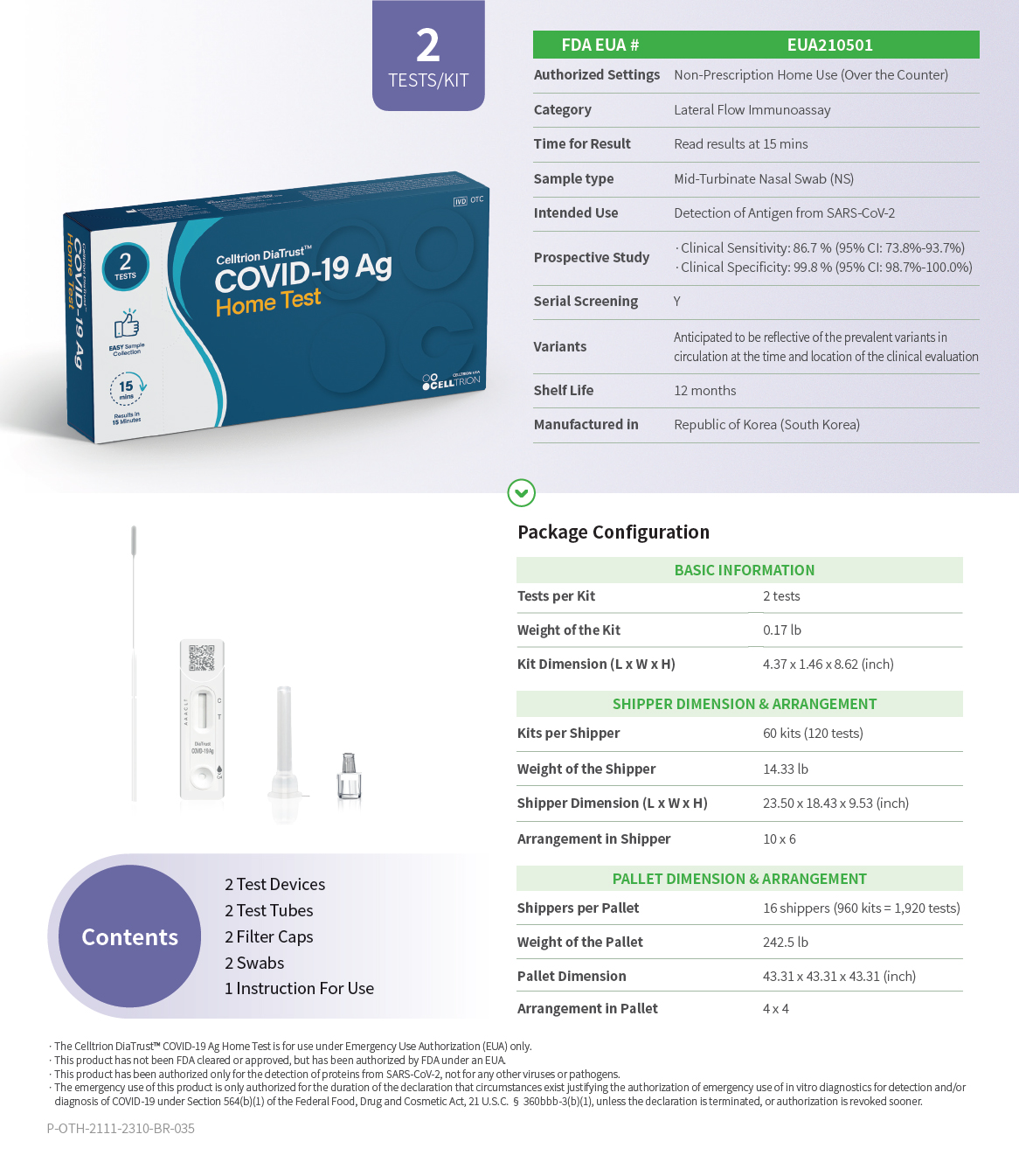
Celltrion DiaTrust™ COVID-19 Ag Home Test

Celltrion DiaTrust™ COVID-19 Ag Rapid Test

